Exabis Library
Welcome to the e-CCO Library!
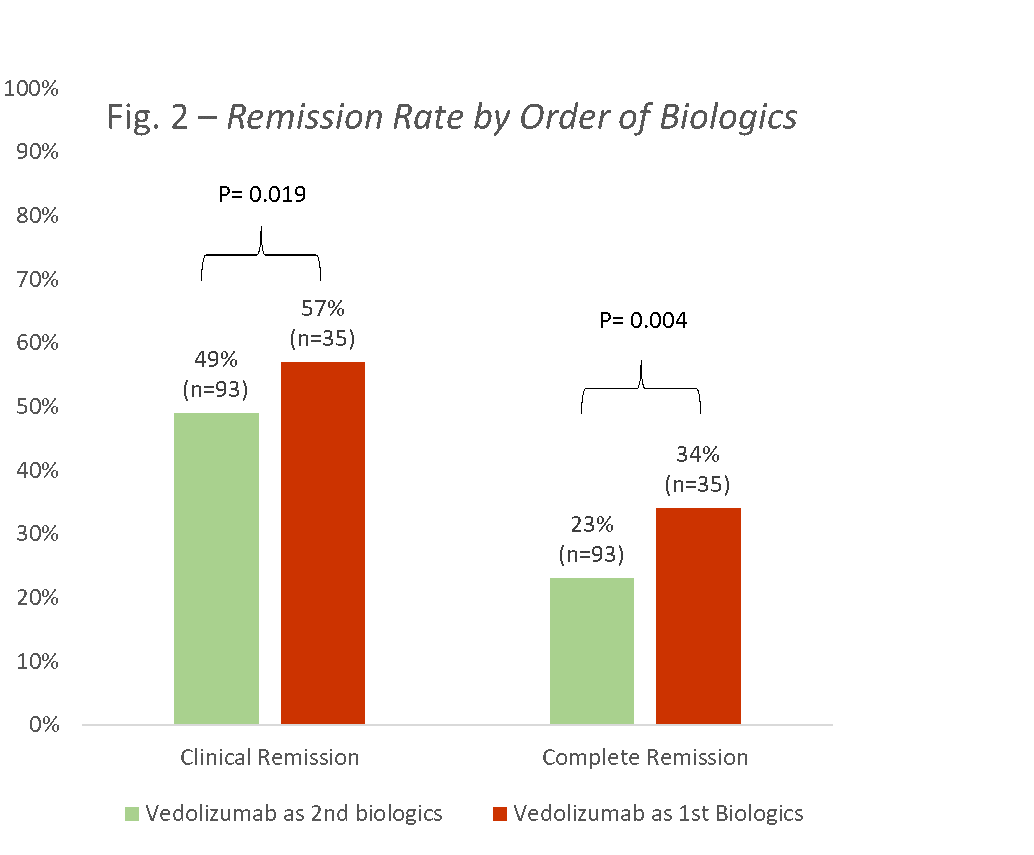
Outcome of induction therapy with vedolizumab in children: Results from the prospective, multi-centre VEDOKIDS study
2022
ECCO'22 Virtual
Tuesday, 24 May 2022, 8:13 PM
Outcomes from the 2015-2016 Fellowships: Hypoxia, antophagy and inflammasome
2017
ECCO'17 Barcelona
Wednesday, 15 March 2017, 2:35 PM by ECCO Administrator
1
Outcomes from the 2015-2016 Fellowships: Post-genomic application in general, and the establishment of computational approaches relevant to translational medicine
2017
ECCO'17 Barcelona
Wednesday, 15 March 2017, 2:44 PM by ECCO Administrator
1
Outcomes from the ECCO Fellowship 2018: Monocyte-derived macrophages as crucial players in the resolution of inflammation and tissue repair in Inflammatory Bowel Disease
2019
ECCO'19 Copenhagen
Tuesday, 28 May 2019, 3:32 PM
1
Outcomes from the ECCO Fellowship 2018: Monocyte-derived macrophages as crucial players in the resolution of inflammation and tissue repair in Inflammatory Bowel Disease
2019
Scientific Programme
Wednesday, 5 June 2019, 9:01 PM
Outcomes from the ECCO Fellowship 2018: Role of TNF-inducible gene-6 (TSG-6) in fistulising Crohn's Disease
2019
ECCO'19 Copenhagen
Tuesday, 28 May 2019, 3:32 PM
1
Outcomes from the ECCO Fellowship 2018: Role of TNF-inducible gene-6 (TSG-6) in fistulising Crohn's Disease
2019
Scientific Programme
Wednesday, 5 June 2019, 9:01 PM
Outcomes from the ECCO Pioneer Award 2017: Faecal transplantation using a novel conditioning method for donor and recipient in mild to moderate treatment refractory colitis in Inflammatory Bowel Disease
2019
ECCO'19 Copenhagen
Tuesday, 28 May 2019, 3:32 PM
1
Outcomes from the ECCO Pioneer Award 2017: Faecal transplantation using a novel conditioning method for donor and recipient in mild to moderate treatment refractory colitis in Inflammatory Bowel Disease
2019
Scientific Programme
Wednesday, 5 June 2019, 9:01 PM
Outcomes meaningful to patients in IBD drug development
2020
5th EpiCom Workshop
Tuesday, 23 June 2020, 4:58 PM
1
Outcomes of first admission for acute severe ulcerative colitis over a decade in England
2019
JCC Podcast
Friday, 28 February 2020, 1:37 PM by Dauren Ramankulov
Tuesday, 13 October 2020, 3:46 PM by Dauren Ramankulov
Outcomes of the ECCO-IOIBD Fellowship 2017: Precision medicine for IBD using advanced machine learning
2019
ECCO'19 Copenhagen
Tuesday, 28 May 2019, 3:32 PM
Outcomes of the ECCO-IOIBD Fellowship 2018 Deciphering the gut-specific B cell response in Inflammatory Bowel Disease
2019
ECCO'19 Copenhagen
Tuesday, 28 May 2019, 3:32 PM
1
Outcomes of the ECCO-IOIBD Fellowship 2018 Deciphering the gut-specific B cell response in Inflammatory Bowel Disease
2019
Scientific Programme
Wednesday, 5 June 2019, 9:01 PM
Overcoming challenges with your ethical committee and GDPR
2020
7th ClinCom Workshop
Tuesday, 23 June 2020, 4:58 PM
1
P0001: Single-cell profiling reveals distinct γδ T cell subsets with protective and pathogenic roles in Ulcerative Colitis
2025
ECCO'25
Monday, 10 February 2025, 2:00 PM
P0002: Identification of Infliximab and Adalimumab -derived peptides using MHC class II antibody mediated immunopeptidomics and mass spectrometry: implications for drug re-design.
2025
ECCO'25
Monday, 10 February 2025, 2:00 PM
P0003: Differential Effects of JAK Inhibitors on Epithelial-Immune Interaction in Inflammatory Bowel Disease
2025
ECCO'25
Monday, 10 February 2025, 2:00 PM
P0004: Transcriptomic profiling as a tool for prediction of endoscopic inflammation in quiescent Crohn's Disease patients
2025
ECCO'25
Monday, 10 February 2025, 2:00 PM
P0005: Transcriptomic analysis reveals molecular differences between intestinal tuberculosis and ileal Crohn's Disease
2025
ECCO'25
Monday, 10 February 2025, 2:00 PM