Exabis Library
Welcome to the e-CCO Library!
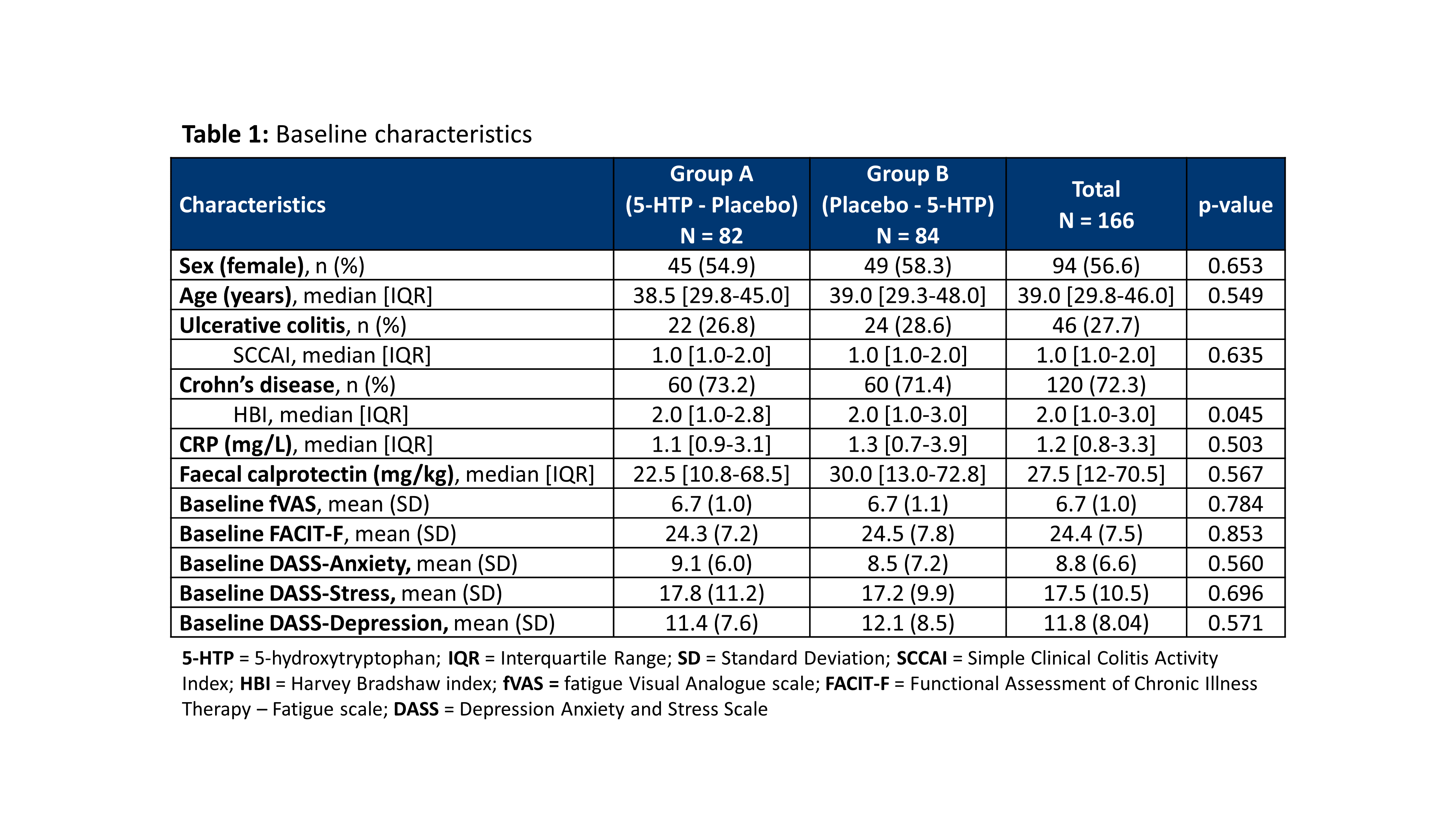
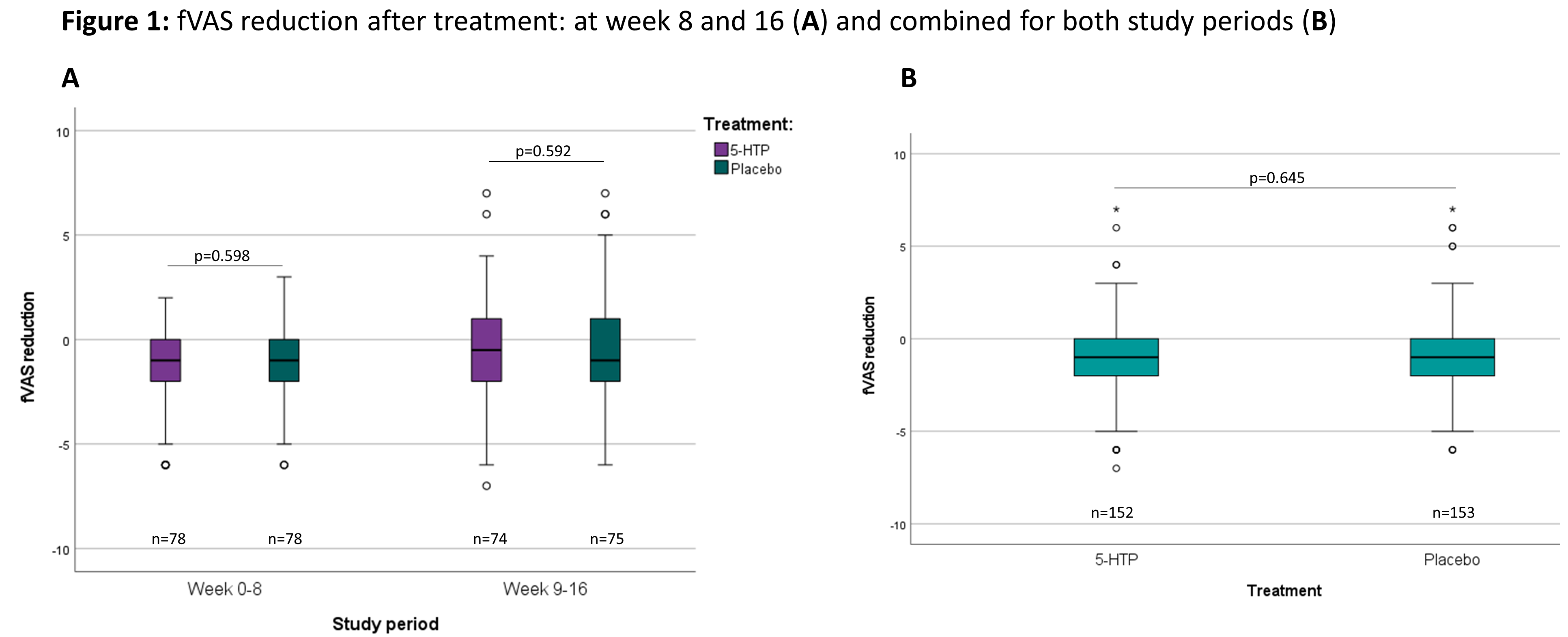
A randomized placebo controlled clinical trial with 5-hydroxytryptophan in patients with quiescent Inflammatory Bowel Disease and fatigue (Trp-IBD)
2022
ECCO'22 Virtual
Tuesday, 24 May 2022, 8:13 PM
A rectovaginal fistula complicating CD
2019
8th S-ECCO IBD Masterclass
Wednesday, 5 June 2019, 9:01 PM
A refractory proctitis complicating CD
2019
8th S-ECCO IBD Masterclass
Wednesday, 5 June 2019, 9:01 PM
A systematic review of the impact of Inflammatory Bowel Disease (IBD) on family members
2022
16th N-ECCO Network Meeting
Tuesday, 24 May 2022, 8:13 PM
Abstract 1: Iterative ileocolonic resection for Crohn's Disease: A prospective multicentric cohort study of the GETAID Chirurgie
2019
8th S-ECCO IBD Masterclass
Wednesday, 5 June 2019, 9:01 PM
Abstract 2: Treatment of perianal fistulas in Crohn’s disease, seton versus anti-TNF versus surgical closure following anti-TNF (PISA): a randomised controlled trial
2019
8th S-ECCO IBD Masterclass
Wednesday, 5 June 2019, 9:01 PM
Abstract 3: Long-term function after transanal versus transabdominal ileal pouch-anal anastomosis for ulcerative colitis: a multicenter cohort study
2019
8th S-ECCO IBD Masterclass
Wednesday, 5 June 2019, 9:01 PM
Abstract presentation: Development of human embryonic stem cell-derived intestinal organoids for in vitro studies on intestinal inflammation and fibrosis
2021
7th Y-ECCO Basic Science Workshop
Friday, 1 October 2021, 12:41 PM
Abstract presentation: Effects of exposure to steroids on the PredictSURE whole blood prognostic assay in Inflammatory Bowel Disease
2021
7th Y-ECCO Basic Science Workshop
Friday, 1 October 2021, 12:41 PM
Abstract presentation: Endoscopically injected allogeneic mesenchymal stromal cells alter the mucosal immune cell compartment in patients with ulcerative proctitis
2021
7th Y-ECCO Basic Science Workshop
Friday, 1 October 2021, 12:41 PM
Abstract presentation: Food-related quality of life in adults with Inflammatory Bowel Disease is influenced by restrictive eating behaviour, disease activity and surgery: a prospective multi-centre study.
2021
6th D-ECCO Workshop
Friday, 1 October 2021, 12:41 PM
Abstract presentation: Risk of eating disorders in Italian children and adolescents with Inflammatory Bowel Disease: results of a multicenter nationwide study
2021
6th D-ECCO Workshop
Friday, 1 October 2021, 12:41 PM
Abstract presentation: Serum proteomics predict endoscopic remission in patients with Crohn’s disease
2021
7th Y-ECCO Basic Science Workshop
Friday, 1 October 2021, 12:41 PM
Abstract presentation: Ulcerative colitis associated single nucleotide polymorphisms found in transcription factor binding sites effect key pathogenesis pathways and facilitate patient stratification
2021
7th Y-ECCO Basic Science Workshop
Friday, 1 October 2021, 12:41 PM
Abstract presentation: αEβ7 positive γδ T cells are associated with mucosal healing in ulcerative colitis and have a homeostatic immunophenotype
2021
7th Y-ECCO Basic Science Workshop
Friday, 1 October 2021, 12:41 PM
Achievement of steroid-free remission in patients with moderately to severely active Crohn’s Disease during treatment with risankizumab
2022
ECCO'22 Virtual
Tuesday, 24 May 2022, 8:13 PM
Achieving high performance in IBD surgery
2021
10th S-ECCO IBD Masterclass
Friday, 1 October 2021, 12:41 PM
Active CD: what should we advise our patients to eat?
2021
Talking Heads
Wednesday, 10 March 2021, 1:57 PM by Dauren Ramankulov
Adalimumab
2020
Educational Audio Podcast
Monday, 27 July 2020, 11:58 AM by Dauren Ramankulov
Monday, 17 August 2020, 10:53 AM by Dauren Ramankulov