Exabis Library
Welcome to the e-CCO Library!
Comparative effectiveness of vedolizumab and ustekinumab in Crohn’s Disease patients who failed anti-TNF treatment: Interrogating 1019 patients from the UK IBD BioResource
2022
ECCO'22 Virtual
Tuesday, 24 May 2022, 8:13 PM
Comparative efficacy of biologics for endoscopic healing of the ileum and colon in Crohn’s Disease
2022
ECCO'22 Virtual
Tuesday, 24 May 2022, 8:13 PM
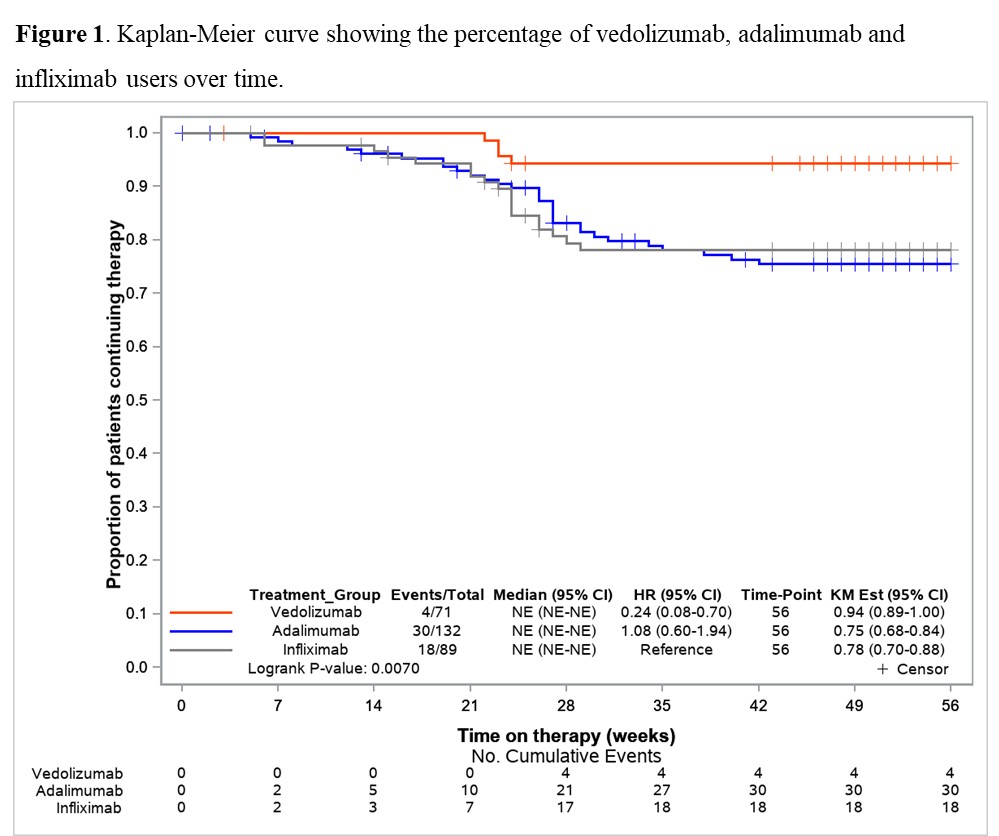
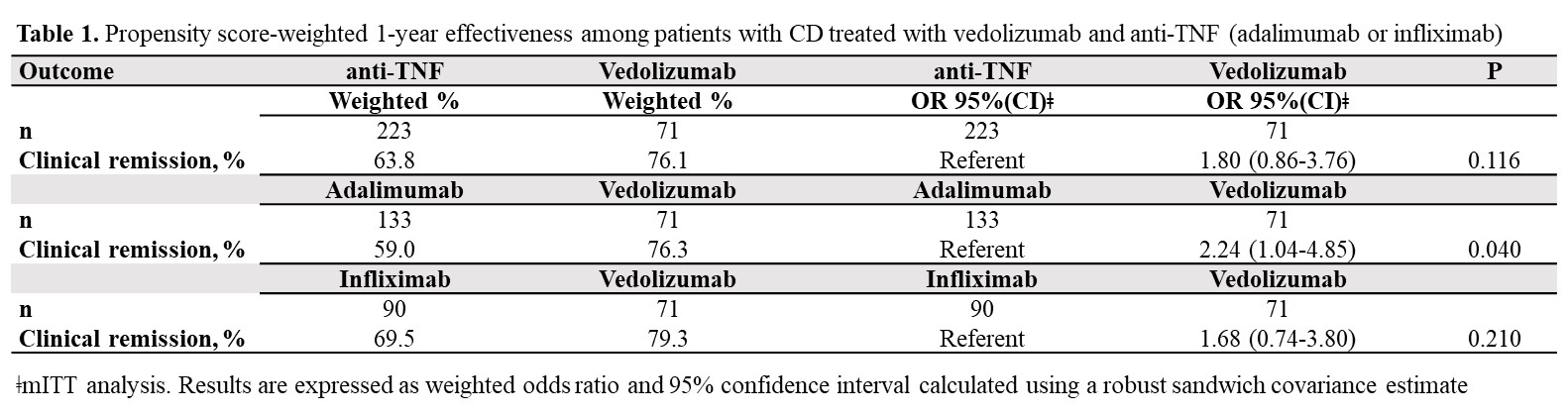
Comparative real-world effectiveness and persistence of vedolizumab versus anti-TNF therapy in biologic-naïve patients with Crohn´s Disease with Propensity Score adjustment: Maintenance phase results at week-52 from the prospective VEDOIBD study
2022
ECCO'22 Virtual
Tuesday, 24 May 2022, 8:13 PM
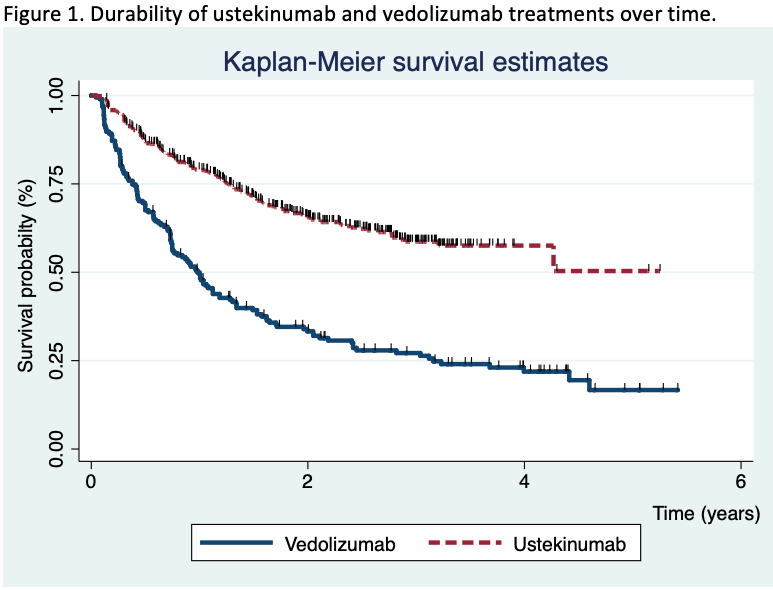
Comparative study of the effectiveness of vedolizumab versus ustekinumab after anti-TNF failure (VERSUS-CD)
2022
ECCO'22 Virtual
Tuesday, 24 May 2022, 8:13 PM
Comparison of the risk of clinical recurrence after ileocolonic resection for Crohn's Disease for modified Rutgeerts' score i2a and i2b categories: Individual patient data meta-analysis
2022
ECCO'22 Virtual
Tuesday, 24 May 2022, 8:13 PM
Complications associated with anti-TNF therapy
2022
20th IBD Intensive Course for Trainees
Tuesday, 24 May 2022, 8:13 PM
Complications secondary to intravenous iron infusion in PIBD patient
2019
4th D-ECCO Workshop
Wednesday, 5 June 2019, 9:01 PM
Computer-aided endoscopy for scoring histological remission in UC
2022
7th H-ECCO IBD Masterclass
Tuesday, 24 May 2022, 8:13 PM
Contemporary nutritional approach in adult IBD (Tandem Talk)
2021
8th P-ECCO Educational Course
Friday, 1 October 2021, 12:41 PM
Context-dependent roles of High-mobility group box 1 (HMGB1) during intestinal inflammation and carcinogenesis
2022
ECCO'22 Virtual
Tuesday, 24 May 2022, 8:13 PM
Contrast enhanced ultrasound and elastography for IBD monitoring?
2021
8th ECCO Ultrasound Workshop - Advanced in collaboration with ESGAR
Friday, 1 October 2021, 12:41 PM
Core outcomes of relevance to prognosis (Position Statement)
2022
6th EpiCom Workshop
Tuesday, 24 May 2022, 8:13 PM
COVID-19 vaccine effectiveness in Inflammatory Bowel Disease patients on tumor-necrosis factor inhibitors: Real world data from a mass-vaccination campaign
2022
ECCO'22 Virtual
Tuesday, 24 May 2022, 8:13 PM
COVID-19 vaccine-induced antibody responses are impaired in Inflammatory Bowel Disease patients treated with infliximab, ustekinumab or tofacitinib, but not thiopurines or vedolizumab
2022
ECCO'22 Virtual
Tuesday, 24 May 2022, 8:13 PM
Crohn’s and the Black death: an evolutionary balance?
2019
JCC Podcast
Monday, 2 September 2019, 4:01 PM by Dauren Ramankulov
Friday, 28 February 2020, 1:33 PM by Dauren Ramankulov
Current knowledge: What is the problem with FMT therapy?
2021
ECCO'21 Virtual
Friday, 1 October 2021, 12:41 PM






 Conclusion
Conclusion